WHAT ARE THE MECHANISMS THAT ENSURE FIDELITY DURING MEIOTIC AND MITOTIC CHROMOSOME SEGREGATION?
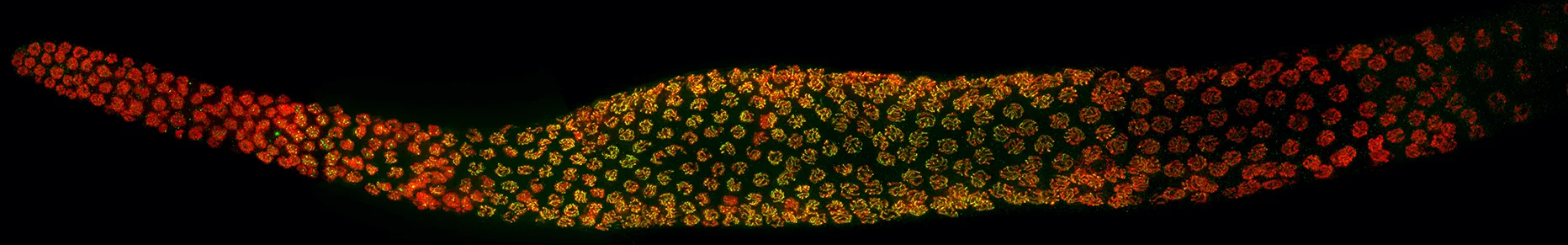
C. elegans germline undergoing meiosis stained to visualize DNA (red) and the protein PCH-2 (green)
MEIOSIS
Cartoon of meiosis
Each person has two versions of their chromosomes: one they received from their mother's egg and one that they received from their father's sperm. These two versions are called homologous chromosomes. Eggs and sperm, also called gametes, are produced by a specialized cell division called meiosis that is essential for sexual reproduction. Meiosis generates haploid gametes from a diploid cell by undertaking two rounds of chromosome segregation: meiosis I (MI above), in which homologous chromosomes segregate, and meiosis II (MII, above), in which sister chromatids segregate.
Upon entry into meiosis, homologous chromosomes replicate to produce sister chromatids. They then pair, synapse and recombine in meiotic prophase (see cartoon above). These events ensure that chromosomes segregate properly to produce eggs and sperm with the correct number of chromosomes. We are interested in understanding how these processes are regulated to avoid the production of aneuploid gametes. Some questions we hope to answer are:
How are the events of pairing, synapsis and recombination monitored during meiotic prophase? If defects occur, what is the response?
How are the events of pairing, synapsis and recombination coordinated during meiotic prophase?
What are the mechanisms that ensure the fidelity of pairing, synapsis and meiotic recombination during meiotic prophase?
MITOSIS
C. elegans two cell embryo in which DNA (red) and the cell membrane (green) can be visualized
During embryonic development, cells rapidly undergo mitosis to generate all the cells of the embryo. These cell divisions are coordinated with cell fate decisions that determine which cells will give rise to which tissues in the adult, indicating an important interplay between developmental fate and control of the cell cycle. The spindle checkpoint is a surveillance mechanism that monitors chromosome segregation to ensure that cells receive the correct number of chromosomes with every cell division. We are interested in understanding how the strength and function of the spindle checkpoint may be modified during development. Some questions we hope to answer are:
What mechanisms ensure a robust spindle checkpoint in large embryonic cells?
What mechanisms ensure a robust spindle checkpoint in immortal germline cells?
Does the requirement for these mechanisms change with the progress of embryonic development?